Tetra Bio-Pharma Provides Update on Clinical Development Program for QIXLEEF™
Tetra Bio-Pharma Provides Update on Clinical Development Program for QIXLEEF™
PR Newswire
OTTAWA, ON, Oct. 14, 2021
OTTAWA, ON, Oct. 14, 2021 /PRNewswire/ - Tetra Bio-Pharma Inc. ("Tetra" or the "Company") (TSX: TBP) (OTCQB: TBPMF) (FRA: JAM1), a leader in cannabinoid-derived drug discovery and development today provided an update on the progress and significant achievements of its lead investigational drug, QIXLEEF™.
QIXLEEF™ is a botanical inhaled drug product with a fixed ratio of THC and CBD that meets USA cGMP regulatory requirements. The drug will be first indicated in patients with cancer suffering of uncontrolled breakthrough pain (REBORN© trials) then in patients with advanced cancer with inadequately controlled pain (PLENITUDE© trials). QIXLEEF™, with its innovative and proprietary dosing data, provides fast acting relief from pain, offering patients a viable, safer, and non-opioid option for pain management.
The Company began regulatory discussions on QIXLEEF™ with both the U.S. Food and Drug Administration (FDA) and Health Canada in mid-2016. This included obtaining a product jurisdiction decision for the regulation of QIXLEEF™ as a drug-device combination product. This type of decision is binding and defines the regulatory path to marketing approval for a combination product. Pre-Investigational New Drug (pre-IND or Type B meetings) and pre-Clinical Trial Application type meetings were held with the regulators to discuss the regulatory requirements to bring QIXLEEF™ to the market as a prescription drug and seek guidance and approval on the drug development strategy.
Since the beginning of the clinical development program in late-2016, QIXLEEF™ strictly adhered to and complied with the regulatory requirements of a prescription drug. The Company completed two Phase 1 clinical trials in healthy volunteers to assess the safety profile, including cardiovascular, pharmacodynamics and pharmacokinetics, of smoked and vaporized QIXLEEF™.
Results of these studies demonstrated the safety of QIXLEEF™ and addressed critical concerns such as the potential for QT prolongation when used in combination with other medications. For patients, the 7-day multiple dosing studies revealed that QIXLEEF™ could be used without functionally impairing the human subject.
From an efficacy point of view, the pharmacodynamic and pharmacokinetic studies demonstrated that QIXLEEF™ could deliver significantly high levels of cannabinoids to the brain within the first 5 minutes of initiating inhalation. The pharmacodynamics confirmed the desired target response in human subjects. The Company believes that these two characteristics of its drug-device combination product are critical for an effective drug in the management of breakthrough pain.
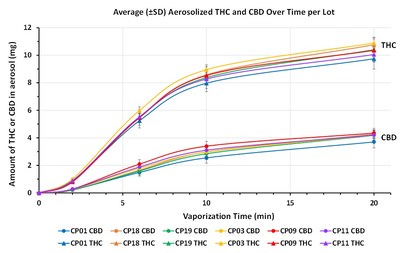
In parallel to the above Phase 1 studies, the Company initiated a major research program on the qualitative and quantitative composition of the aerosols generated by smoking and vaporizing QIXLEEF™. This research began in mid-2017 and continues to be part of the Company's routine analysis of QIXLEEF™ and its raw materials to ensure consistent delivery of cannabinoids to the patients between production lots. This research program led to the development of composition of matter intellectual property.
In November 2019, the Company announced that the FDA had authorized the initiation of our PLENITUDE© trial after demonstrating new quality standards for mitigating the risk of mycotoxins to patients. PLENITUDE© is a 4-week double-blind, randomized, placebo-controlled trial to evaluate the safety and efficacy of inhaled QIXLEEF™ on uncontrolled pain related to cancer in 78 adult patients with advanced incurable cancer. The Company received the raw materials for fabricating QIXLEEF™ in September 2020 thereby allowing the start of the in-life phase of the trial.
In August 2020, the Company shared data on its CBD Metabolite Study in Humans. This milestone had a major positive impact on Tetra's inhaled product portfolio, as the data demonstrated very low level of CBD metabolites in humans exposed to inhaled QIXLEEF™. This evidence corroborated the absence of liver toxicity when QIXLEEF™ is inhaled and strengthened the safety profile of our drug product.
In 2020, the Company expanded its regulatory activities to ensure the nonclinical safety strategy, manufacturing and quality, and a global clinical trial program for the marketing approval of QIXLEEF™ in Europe. Subsequently, Tetra created a subsidiary in Europe, called Tetra Bio-Pharma Europe LTD. The former was important so that the regulatory requirements for QIXLEEF™ would be similar for Europe, United States and Canada. Having a global drug development strategy ensures economy of scale and lower cost of drug development to shareholders. As part of the regulatory achievements, in September 2021, the Company received its first Scientific Advice Assessment Report from a European Medicines Authority. This Report provided positive feedback on the drug development plan and eligibility for submitting a Marketing Authorization Application (MAA) under Directive 2001/83/EC (Directive).
In January 2021, the FDA authorized the initiation of the REBORN1© trial with QIXLEEF™. The REBORN1© trial is a head-to-head phase 2 study against an opioid treatment in the management of short and frequent episodes of incapacitating pain (breakthrough pain) requesting immediate release opioid treatment in patients living with cancer. The initial protocol was assessing a direct safety and efficacy comparison between QIXLEEF™ and oral morphine sulfate immediate release on the onset of pain relief in this population. In June 2021, an approved amended protocol opened the recruitment to cancer patients with breakthrough pain who are treated with either oral morphine sulfate immediate release, oral hydromorphone immediate release, or oral oxycodone immediate release. REBORN1©, if successful, is expected to provide proof of concept evidence that QIXLEEF™ can be an alternative to the class of immediate release oral opioids in the management of breakthrough cancer pain.
With a supportive outcome of the REBORN1© trial, the Company will be on a path to bring QIXLEEF™ as a novel, safe, and effective treatment for the management of breakthrough pain. The Company will leverage the long-term safety data gathered in the PLENITUDE© trial to supplement the safety and efficacy data for the REBORN© trials. The REBORN© clinical strategy provides the fastest and lowest cost development program to bring QIXLEEF™ to the market.
The Company is finalizing its raw material global supply plans and is in partnering discussions as it prepares to initiate the last stage of its drug development of QIXLEEF™ for an indication in breakthrough pain as an alternative to immediate-release oral opioids.
Dr. Guy Chamberland, CEO and CRO commented, "Our team has made significant progress in bringing QIXLEEF™ to a late clinical development stage. We are dedicated to bringing this novel medication to patients and physicians as an alternative to opioids. It has been a challenging, but exciting journey to develop the first Rx inhaled cannabinoid-based drug."
About Tetra Bio-Pharma
Tetra Bio-Pharma (TSX: TBP) (OTCQB: TBPMF) (FRA:JAM1) is a leader in cannabinoid-derived drug discovery and development with a FDA and a Health Canada cleared clinical program aimed at bringing novel prescription drugs and treatments to patients and their healthcare providers. Our evidence-based scientific approach has enabled us to develop a pipeline of cannabinoid-based drug products for a range of medical conditions, including pain, inflammation, and oncology. With patients at the core of what we do, Tetra Bio-Pharma is focused on providing rigorous scientific validation and safety data required for inclusion into the existing biopharma industry by regulators, physicians and insurance companies.
For more information visit: www.tetrabiopharma.com
Neither the TSX Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Exchange) accepts responsibility for the adequacy or accuracy of this release.
Forward-looking statements
Some statements in this release may contain forward-looking information. All statements, other than of historical fact, that address activities, events or developments that the Company believes, expects or anticipates will or may occur in the future (including, without limitation, statements regarding potential acquisitions and financings) are forward-looking statements. Forward-looking statements are generally identifiable by use of the words "may", "will", "should", "continue", "expect", "anticipate", "estimate", "believe", "intend", "plan" or "project" or the negative of these words or other variations on these words or comparable terminology. Forward-looking statements are subject to a number of risks and uncertainties, many of which are beyond the Company's ability to control or predict, that may cause the actual results of the Company to differ materially from those discussed in the forward-looking statements. Factors that could cause actual results or events to differ materially from current expectations include, among other things, without limitation, the inability of the Company to obtain sufficient financing to execute the Company's business plan; competition; regulation and anticipated and unanticipated costs and delays, the success of the Company's research and development strategies, including the success of this product or any other product, the applicability of the discoveries made therein, the successful and timely completion and uncertainties related to the regulatory process, the timing of clinical trials, the timing and outcomes of regulatory or intellectual property decisions and other risks disclosed in the Company's public disclosure record on file with the relevant securities regulatory authorities. Although the Company has attempted to identify important factors that could cause actual results or events to differ materially from those described in forward-looking statements, there may be other factors that cause results or events not to be as anticipated, estimated or intended. Readers should not place undue reliance on forward-looking statements. The forward-looking statements included in this news release are made as of the date of this news release and the Company does not undertake an obligation to publicly update such forward-looking statements to reflect new information, subsequent events or otherwise unless required by applicable securities legislation.
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/tetra-bio-pharma-provides-update-on-clinical-development-program-for-qixleef-301400196.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/tetra-bio-pharma-provides-update-on-clinical-development-program-for-qixleef-301400196.html
SOURCE Tetra Bio-Pharma Inc.